**The effective date for the updates to the Notifiable Conditions regulations in chapter 246-101 WAC has been postponed until January 1, 2023. Please click here to learn more about the postponement.**
| To: | Hospital Chief Information Officers, Chief Medical Officers, Chief Quality Officers, Laboratory Directors, Legal Counsel, and Government Affairs Staff |
| Staff Contact: | David Streeter, Policy Director- Clinical and Data DavidS@wsha.org | (206) 216-2508 |
| Subject: | Beginning January 1, 2023 – New Data Requirements for Revised Notifiable Conditions Regulations |
Purpose
The purpose of this bulletin is to inform hospitals about revisions to Washington State’s Notifiable Conditions regulations in chapter 246-101 WAC that take effect January 1, 2023. The regulatory revisions:
- Require health care facilities, health care providers, and laboratories to collect extensive patient race, language, and ethnicity data;
- Clarify the reporting process for conditions deemed notifiable, require reporting through secure electronic data transmission, and eliminate phone calls as a reporting method; and
- Add 21 new conditions to the list of conditions that are deemed notifiable to local health jurisdictions (LHJ) and the Department of Health (DOH).
Hospitals should begin preparing to comply with the revised regulations in advance of their effective date. Preparations may include developing new procedures for collecting and reporting the expanded patient demographic data and adjusting electronic health record systems (EHR) to store the additional patient data.
WSHA provided member feedback to the Washington State Board of Health (WSBOH) during the regulatory process. WSHA’s feedback resulted in the January 1, 2023 effective date instead of an earlier date. DOH anticipates its’ technical work to implement the regulations will be complete in advance of the January 1, 2023 effective date, and WSHA will continue to seek status updates from DOH. Hospitals should watch for updates from WSBOH, DOH, and WSHA as the effective date for the revised regulations gets closer.
The new notifiable conditions rules are separate from the requirements in HB 1272 (2021) that take effect in 2023.
Recommendations
- Review this bulletin and chapter 246-101 WAC to understand the forthcoming changes to Washington State’s Notifiable Conditions regulations.
- Share this bulletin with relevant staff so they are aware of the forthcoming changes.
- Plan for changes to your patient data collection process and data reporting workflow to comply with the new requirements.
Applicability/Scope
The updated notifiable conditions rules apply to:
- Health care facilities including hospitals licensed under chapter 70.41 RCW, ambulatory surgical centers licensed under chapter 70.230 WAC, and “Clinics, or other settings where one or more health care providers practice,” among other facilities;
- Health care providers licensed under Title 18 RCW and “Military personnel providing health care within the state regardless of licensure;” and
- Laboratories licensed under chapter 70.42 RCW.
Please see the definitions of “health care facility,” “health care provider,” and “laboratory” in revised WAC 246-101-010 for the full list of facilities and individuals subject to the regulations.
Additionally, revised WAC 246-101-105 permits health care facilities to assume the reporting responsibilities for a health care provider practicing within the facility. However, health care facilities remain prohibited from assuming the reporting responsibilities for laboratories that are components of their facilities.
Overview
Washington State’s Notifiable Conditions regulations list the health conditions that must be reported to LHJs and DOH; the information that is reported; and the reporting process. Currently:
- Most hospitals report notifiable conditions as electronic case reports (ECR) by fax to DOH and by fax or phone to LHJs because an electronic method is currently unavailable. DOH’s Notifiable Conditions website contains the ECR forms.
- Most laboratories report electronic laboratory reports (ELR) electronically through their EHR systems or are in process of transitioning from fax to ELR reporting. ELRs are submitted via OneHealthPort using HL7, which is an electronic language for transferring information.
The current rules took effect in 2011 and have not been updated until now. Because of this, the current regulations omit new conditions identified by WSBOH as warranting notification to LHJs and DOH. The current regulations also do not officially require reporting entities to collect patient demographic information. WSBOH addressed these issues in the updated regulations that take effect January 1, 2023.
New Race, Ethnicity, and Language Data Requirements
The current Notifiable Conditions regulations in chapter 246-101 WAC do not require health care facilities, health care providers, and laboratories to collect and report patients’ race, ethnicity, and language data. Instead, hospitals, health care facilities, and providers collect and report this information using the fields available on DOH’s ECR forms, while laboratories report through the ELR process. The WAC’s silence creates challenges for tracking conditions across populations, which can make it more difficult to respond to affected communities. To remedy this, the updated regulations require health care providers, health care facilities, and laboratories to collect demographic information from patients and report it with each notifiable condition case. This new requirement will provide public health agencies with information that can help target responses to disease outbreaks that impact specific racial and ethnic communities.
New WAC 246-101-011 specifies the different options patients may choose when asked about their race, ethnicity, and preferred language.
- Ethnicity: Patients can identify as (a) Hispanic, Latino/a, Latinx or (b) Non-Hispanic, Latino/a, Latinx.
- Race: Patients may choose from a list of 72 different race options. Most reporting entities are currently collecting seven broad race categories for ECRs and ELRs. The current categories are American Indian/Alaska Native, Asian, Black/African American, Native Hawaiian/Other Pacific Islander, White, Other, and Unknown. However, the regulations expand the current categories into more detailed identities. For example, the Alaska Native/American Indian race option is broken down into separate Alaska Native and American Indian options.
- Language: Patients may choose from a list of 50 language options.
Patients may select “unknown” for all three categories, if appropriate. Importantly, patients may also decline to respond to the demographic questions. In such circumstances, staff members collecting the patient data and preparing the ECR and ELR should select the “Patient declined to respond” option for each category. The new data collection mandates will require changes to hospitals’ existing patient data collection processes to incorporate the demographic questions and response options. DOH will be updating its ECR forms and issuing resources with HL7 ELR specifications to reflect the changes made by WAC 246-101-011. Please see new WAC 246-101-011 for the complete race, ethnicity, and language data requirements.
Reporting Process Changes
The revised rules make several important changes to the notifiable condition reporting process.
- Phone Reporting Discontinued: The option to report notifiable conditions over the phone is removed. Instead, all reports must take place through secure electronic data transmission, which is defined as: “electronic communication and accounts developed and maintained to prevent unauthorized access, loss, or compromise of sensitive information including, but not limited to, secure file transfer, secure email, secure facsimile, a health information exchange authorized under RCW 41.05.039, and secure electronic disease surveillance system.” Laboratories will continue to use the existing ELR process, while health care facilities and providers will continue to fax ECR forms. However, DOH intends to make an electronic submission process available to hospitals and health care facilities in the future.
- Clearer Reporting Tables: The tables in WAC 246-101-101 (applicable to health care facilities and providers) and WAC 246-101-201 (applicable to laboratories) are clarified for easier use. The revised tables show who must report a notifiable health condition; whether the report for each condition is sent to state or local authorities; and the deadline for submitting each condition report.
- Updated Confidentiality Provisions: The current data confidentiality provisions in WAC 246-101-120 and WAC 246-101-220 are simplified to state health care facilities, health care providers, and laboratories “shall maintain the confidentiality of health care information consistent with chapter 70.02 RCW and any other applicable confidentiality laws.” The revision also requires health care facilities, health care providers, and laboratories to “establish and implement policies and procedures to maintain the confidentiality of health care information.”
- Updated Specimen Submission: The specimen submission requirements for laboratories are clarified to reflect the changes made in chapter 246-101 WAC.
- Consolidated WACs: The reporting requirements for health care providers and health care facilities are merged into one standardized set of requirements in WACs 246-101-105, 110, 115, and 120. This change ensures consistency among the reports submitted by providers and facilities. Please see WACs 246-101-105, 110, 115, and 120 to review the standardized process.
New Conditions Deemed Notifiable for Hospitals
The current list of conditions deemed notifiable to DOH and LHJs first took effect in 2011. Since then, new health conditions have been identified as warranting formal notification to DOH and LHJs when cases are identified. Because of this, the updated regulations add COVID-19, specific birth defects, and 21 new health conditions to the list of conditions that are notifiable to DOH and LHJs. The added conditions are:
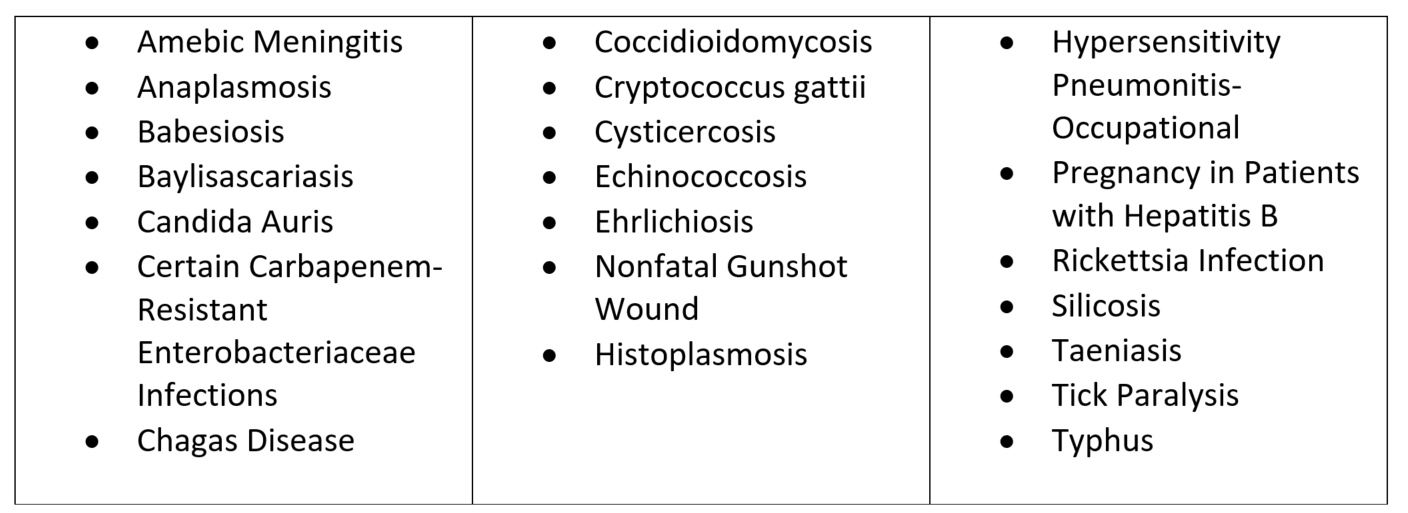
The new regulations also remove three categories of conditions that are reported under the current rules. The removed categories are “Other Rare Diseases of Public Health Significance,” “Emerging Conditions with Outbreak Potential,” and “Disease of Suspected Bioterrorism Origin.”
Please see WAC 246-101-101 for the table containing the conditions hospitals, health care facilities, and health care providers must report; and WAC 246-101-201 for the table containing the conditions laboratories must report.
Next Steps
Hospitals should begin preparing to comply with the revised regulations when they take effect on January 31, 2022. Preparations may include:
- Developing procedures for collecting and reporting the expanded patient data; and
- Adjusting EHR systems to be able to store the additional data collected from patients.
WSHA is currently working with hospitals that are piloting processes to improve the collection of patient demographic data through culturally sensitive methods. As part of that work, WSHA has compiled a list of resources hospitals can use to plan for patient data collection. Please contact Abby Berube for information and additional resources.
DOH does not anticipate advising hospitals on how to develop their internal data collection and reporting procedures.
DOH recently started its implementation work to prepare for the high volume of data it will receive and process. The new regulations require DOH to:
- Configure its’ data systems to collect and store the new data submitted under chapter 246-101 WAC;
- Publish updated ECR forms for hospitals, health care facilities, and providers to use; and
- Provide resources with updated HL7 specifications for ELRs.
DOH is also working to develop a future electronic submission method for ECRs, however the work is delayed. Hospitals seeking more information about the future electronic submission method should contact ECR@doh.wa.gov to learn what modifications they should make to their EHR system to align with DOH’s future electronic reporting system.
DOH anticipates its’ work will be complete by January 31, 2022, and WSHA will continue to seek status updates from DOH. Hospitals should watch for updates from WSBOH, DOH, and WSHA as the effective date for the revised regulations gets closer. Questions about the revised regulations may be submitted via email to notifiableconditions@sboh.wa.gov.
Background
According to WSBOH’s Concise Explanatory Statement, “the purpose of rulemaking within chapter 246-101 WAC is to better protect public health by improving public health authorities’ understanding of emerging conditions, allowing more thorough case investigations, and improving the public health response to infectious and noninfectious conditions.” The final rules issued by WSBOH are the culmination of nearly three years of work to update the regulations, which had not been updated since their enactment in 2011.
WSHA provided member feedback to WSBOH during the rule making process. WSHA recommended WSBOH delay the revised regulations’ implementation to ensure alignment with HB 1272 (2021), which also requires expanded patient demographic information reporting to DOH. This resulted in the January 1, 2023 effective date for the Notifiable Conditions regulations despite later effective dates in HB 1272. WSHA continues to advocate for alignment between the Notifiable Conditions regulations and the forthcoming regulations for HB 1272 to ensure efficiency and consistency between the two data collection mandates.
WSHA’s 2021 New Law Implementation Guide
Please visit WSHA’s new law implementation guide online. The Government Affairs team is hard at work preparing resources and information on the high-priority bills that passed in 2021 to help members implement the new laws, as well as links to resources such as this bulletin. In addition, you will find the Government Affairs team’s schedule for the release of upcoming resources on other laws and additional resources for implementation.
References
Chapter 246-101 WAC– includes the current rules and the revisions effective January 1, 2023
Washington State Board of Health Notifiable Conditions Rules Webpage
Washington State Department of Health Notifiable Conditions Webpage